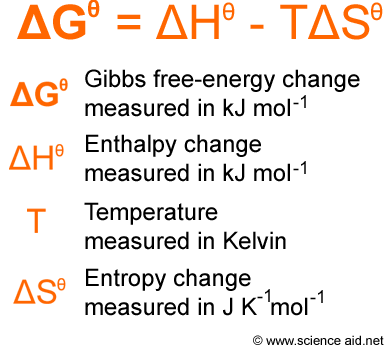3 Laws of thermodynamics
3 laws of thermodynamics
First Law
_Energy is constant
It cannot be created or destroyed - only converted from one form into another.
It cannot be created or destroyed - only converted from one form into another.
Second Law
Every energy transfer increases the entropy of the universe due to the loss of unstable energy.
Any spontaneous process increases the disorder of the universe. (Entropy)
For more information on the second law of thermodynamics visit this website:
https://www.boundless.com/biology/textbooks/boundless-biology-textbook/metabolism-6/potential-kinetic-free-and-activation-energy-69/the-second-law-of-thermodynamics-348-11485/
Any spontaneous process increases the disorder of the universe. (Entropy)
For more information on the second law of thermodynamics visit this website:
https://www.boundless.com/biology/textbooks/boundless-biology-textbook/metabolism-6/potential-kinetic-free-and-activation-energy-69/the-second-law-of-thermodynamics-348-11485/
Entropy
|
Entropy as described by Crash Course.
Watch this video from: 2:01 - 3:27 |
|
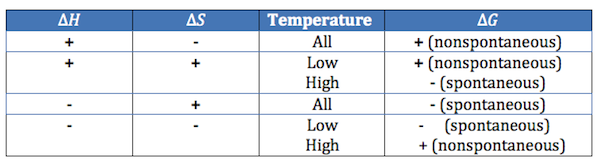
Now that you have looked at this video, here is a table that may help your students determine spontaneity
Gibbs Free energy
Energy that can do useful work!
Positive Gibbs Free Energy (+G): need for energy, a reaction will require an energy input to occur (i.e. non-spontaneous)
Negative Gibbs Free Energy (-G): free energy is being released for the purpose of useful work (i.e. spontaneous)
Positive Gibbs Free Energy (+G): need for energy, a reaction will require an energy input to occur (i.e. non-spontaneous)
Negative Gibbs Free Energy (-G): free energy is being released for the purpose of useful work (i.e. spontaneous)
Third law
At absolute zero temperature: The entropy of a perfect crystal at absolute zero is exactly equal to zero.
This law does not apply to SBI4U as it does not pertain to living organisms.
This law does not apply to SBI4U as it does not pertain to living organisms.