Important Concepts
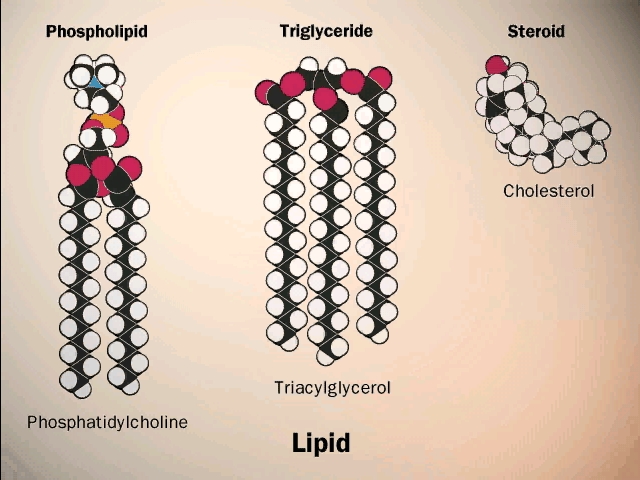
Lipids are hydrophobic (water disliking molecules composed of carbon, hydrogen, and oxygen atoms. They are molecules which contain fewer polar O-H bonds and more non-polar C-H bonds compared to carbohydrates. Lipids are insoluble in water but very soluble in other non-polar substance (remember likes dissolve likes). Organisms use lipids for storing energy, building membranes (using phospholipids), and as chemical signalling molecules. There are 4 families of lipids listed below: fats, phospholipids, steroids, and waxes. Fats are the most common energy-storing molecules in living organisms (more so than carbohydrates). The most common storage of fats in plants and animals is in the form of triglycerols (or triglycerides). Triglycerides contain 3 fatty acids attached to glycerol through a condensation reaction, resulting in a bond known as an ester linkage. This process is known as esterification. The membrane of cells is mainly composed of fats known as phospholipids. Phospholipids are composed of glycerol, two fatty acids and a highly polar phosphate group. Therefore the whole molecule had a polar hydrophilic head and two non-polar hydrophobic tails. This allows the cell membrane to separate two water compartments, such as extracellular fluid and the cells cytoplasm. Membranes contain the third class of lipids known as sterols. Sterols are again hydrophobic molecules which contain 4 fused hydrocarbon rings. Cholesterol is one of the most important steroid components of cell membranes. The forth class of lipids are known as waxes. Waxes contain long-chain fatty acids linked to alcohols or carbon rings. These molecules are hydrophobic with a firm, pliable consistency. These properties allow for waxes to be well suited for waterproof coating plants and animals.
|
Quick Facts
Saturated fatty acids: All single bonds between C atoms, max amount of H’s Unsaturated fatty acids: 1 or more double bonds between C atoms, fewer than max amount of H’s
|
Curriculum Links
B2.3 construct and draw three-dimensional molecular models of important biochemical compounds, including carbohydrates, proteins, lipids, and nucleic acids [PR, C]
B3.2 describe the structure of important biochemical compounds, including carbohydrates, proteins, lipids, and nucleic acids, and explain their function within cells
B3.3 identify common functional groups within biological molecules (e.g., hydroxyl, carbonyl, carboxyl, amino, phosphate), and explain how they contribute to the function of each molecule
B3.2 describe the structure of important biochemical compounds, including carbohydrates, proteins, lipids, and nucleic acids, and explain their function within cells
B3.3 identify common functional groups within biological molecules (e.g., hydroxyl, carbonyl, carboxyl, amino, phosphate), and explain how they contribute to the function of each molecule
Misconceptions
- Most people think that lipids (fats) are only found in common foods such as butter and oil. This however is not that case, and many common foods do contain fats, like egg yolk for example. The food lab below is geared at getting students to identify different types of food that contain lipids to address this misconception. Fats come in many different forms and it is important to teach students how to identify if a food does or does not contain fats.
Activities and Teaching Strategies
|
It is important to show students the structure of triglycerides and phospholipids and the difference between the two. Here is great activity to use in the classroom to introduce students to the different structures of triglycerides and phospohlipids.
It is important to explain the properties of lipids to students and how to identify them in our foods. Here is a great lab to do with your students to allow them to explore different foods and identify if they contain lipids (fats) in them.
|
STSE Application #1
|
Hydrogenation
Hydrogenation is a process that uses hydrogen gas to change liquid vegetable oils into a hard spread/margarine. This process stabilizes the oil and prevents spoilage from oxidation. Hydrogenation reactions are sensitive to a variety of factors that can negatively impact batch time, catalyst life, production rate, and selectivity. This process breaks the double bond found in unsaturated fatty acids and add hydrogens to the carbon chain, and thus resulting in a more solid structure. Therefore the complete hydrogenation of unsaturated fatty acids results in saturated fatty acids. The partial hydrogenation of unsaturated fatty acids results in tans-fats. This is a common process in the food industry today. Changing the degree of saturation of the fat changes some important physical properties, such as the melting range, which is why liquid oils become semi-solid. Solid or semi-solid fats are preferred for baking because the way the fat mixes with flour produces a more desirable texture in the baked product. However, it has been known that trans-fats are not healthy for human consumption and that is why many food now are advertised as being free of trans-fats. |
|
Assessment #2
|
Macromolecule Quiz
|